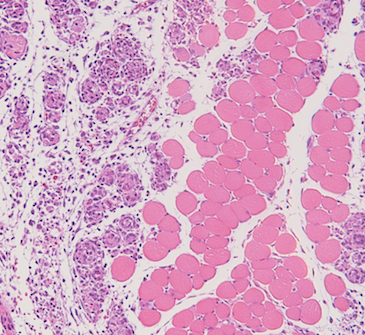
Research
Overview
Our lab is keenly interested in the molecular mechanism of muscle formation or myogenesis. We utilize versatile genetic mouse models, cell culture tools and biochemical approaches to understand the mechanism of muscle development and regeneration.
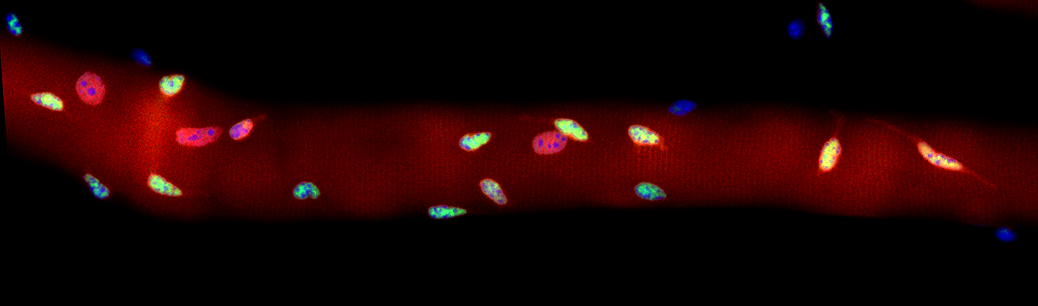
1. Genetic Basis of Muscle Cell Fusion.
The basic cellular unit of skeletal muscles is muscle fiber or myofiber. It transverses nearly the full length of a muscle and can easily reach to many centimeters long. Formation of myofiber depends on the fusion of many hundreds’ to thousands’ regularly sized muscle precursor cells. “Unity makes strength” is perfectly manifested by the muscle cell fusions, which make the synchronized force generation possible. We aim to discover the genetic circuits wired in our muscle cells that control this process.
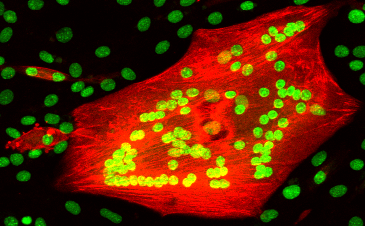
2. Molecular Regulation of Muscle Stem Cell.
Skeletal muscles have remarkable regeneration potentials. The secrets are the stem cells that reside on myofibers. In healthy tissues, muscle stem cells largely remain in dormancy or a quiescent state. But they can respond to myofiber injury promptly, and orderly undergo the processes of proliferation, differentiation and fusion to repair injured muscles. Although working hard, muscle stem cells do not exhaust themselves and know precisely when to self-replicate & return to the quiescence. This self-renewal decision is essential for allowing them to regenerate tissues in the future. We therefore study the key signaling pathways that determine the fate choice of muscle stem cells.
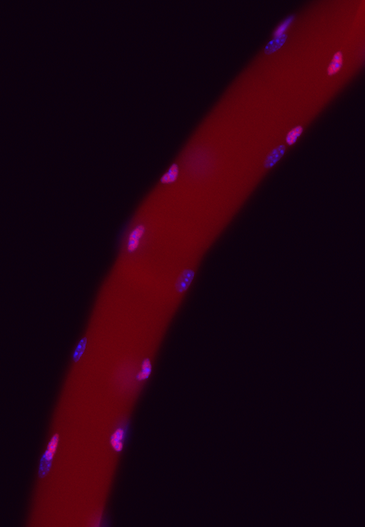
3. Improve Muscle Regeneration.
There are more than 100 types of monogenic muscular dystrophies and myopathies. Among them, the most common and severe one is the Duchenne muscular dystrophy (DMD). Patients with DMD suffer progressive muscle weakness in early ages, lose ambulation in their teens, and ultimately die in the 20–30’s due to respiratory and cardiac function failures. Better treatments of muscle diseases including DMD depends on the deeper understanding of the cellular and molecular mechanism of muscle regeneration. We value the translational potential of our basic biological research and eagerly explore the new strategies that could lead to better therapeutic outcomes of muscle diseases.